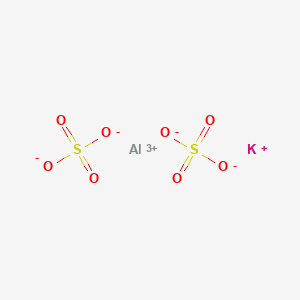
SOLVED: a) Is 12H2O included in calculating molar mass of an alum? Explain. b) Will the water molecules be attached to monovalent or trivalent cation? Explain. c) 1.5 g of aluminum is

High Quality Potash Alum Kal(so4)2.12h2o - Buy High Purity Aluminium Potassium Sulfate,Aluminium Potassium Sulphate For Food Addictive,Food Grade Aluminium Potassium Sulphate Product on Alibaba.com

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

KAl(SO4)2.12H2O (Alum) Catalyzed Organic Synthesis: Patel, Kinjal, Vekariya, Rajesh, Patel, Hitesh: 9783659939518: Amazon.com: Books

Aluminum Potassium Sulphate Kal (so4)2.12h2o Shipment Quickly From China - Buy Aluminum Potassium Sulphate Lowest Price,Aluminum Potassium Sulphate Lower Price,Aluminun Potassium Sulphate Price In China Product on Alibaba.com


















![PDF) Potash alum [KAL(SO4)2.12H2O] catalysed esterification of formylphenoxyaliphatic acids PDF) Potash alum [KAL(SO4)2.12H2O] catalysed esterification of formylphenoxyaliphatic acids](https://i1.rgstatic.net/publication/236840970_Potash_alum_KALSO4212H2O_catalysed_esterification_of_formylphenoxyaliphatic_acids/links/02e7e5194eabc81131000000/largepreview.png)