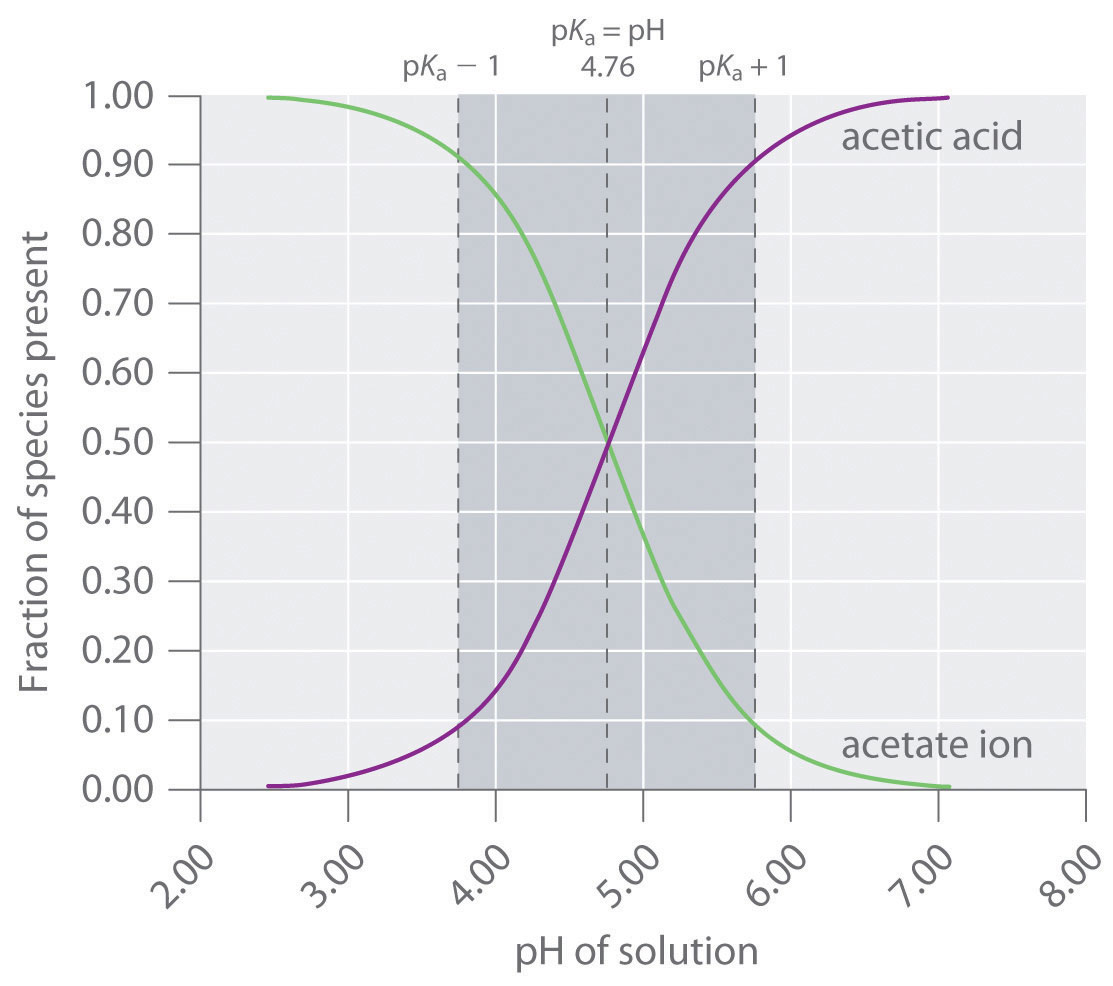
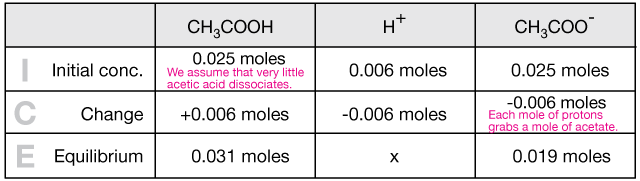
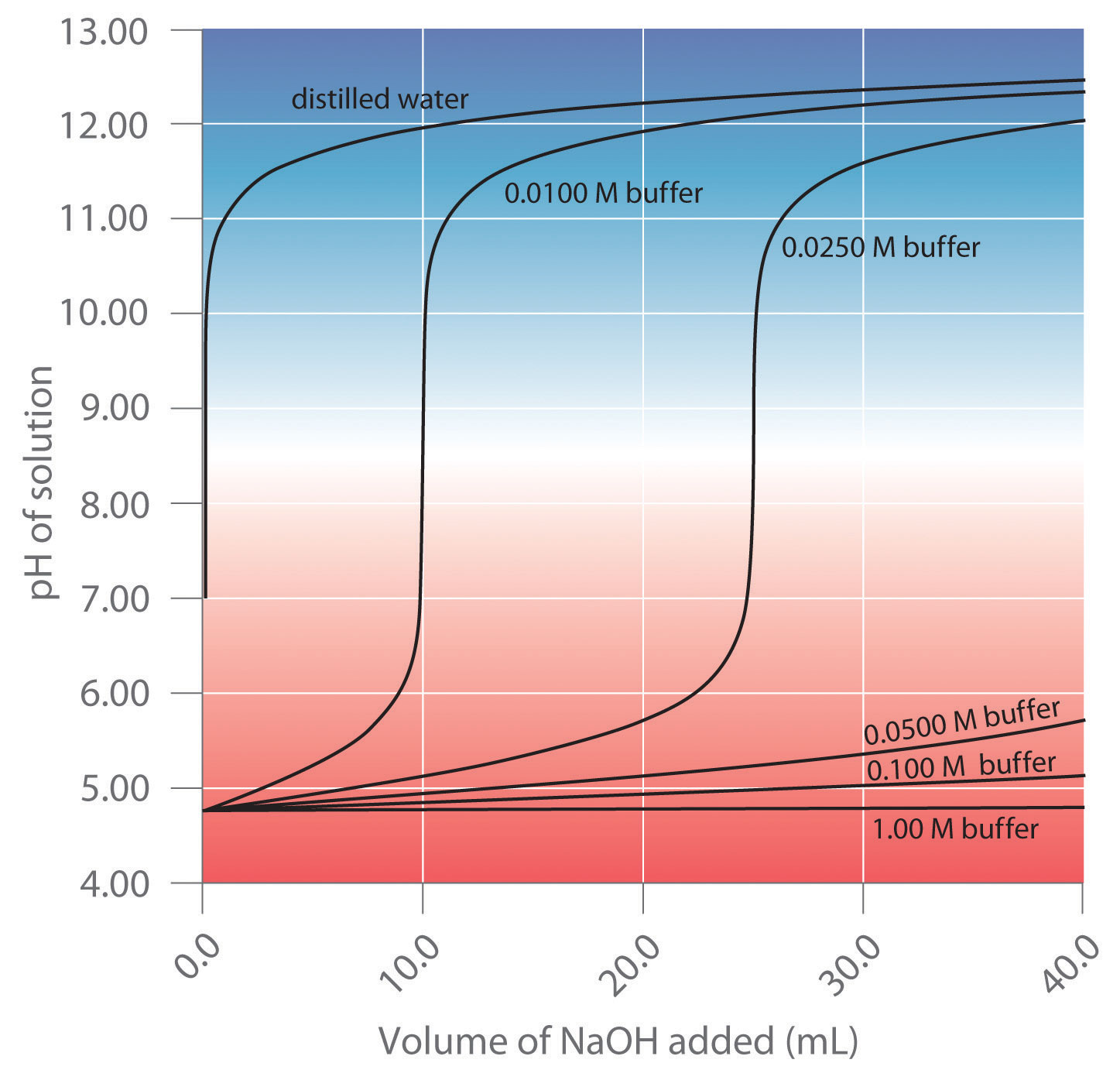
SOLVED:A sodium acetate-acetic acid buffer solution was prepared by adding a 0.020 M̅ HCl solution to 500 mL of 0.020 M CH3 COONa and then diluting the mixed solution to 1.0 L.

E-Lifes: Acetate buffer preparation and calculation. (Weak acid + salt of weak acid , Weak acid + salt of weak acid, Weak acid + strong acid)

Chapter 17 Acid-Base & Solubility Equilibria The Common Ion Effect 17.2 Buffer Solutions 17.3Acid-Base Titrations (omitted) 17.4Solubility Equilibria. - ppt download

Preparation of acetate buffers using Sodium acetate and acetic acid using the Henderson-Hasselbach - Studocu

A buffer solution is prepared by mixing `10ml` of `1.0 M` acetic acid & `20 ml` of `0.5 M` - YouTube
When a small amount of HCL is added to a buffer solution of acetic acid and sodium acetate what happen?
![Calculate pH of a buffer prepared by adding 10 mL of 0.10 M acetic acid to 20 mL of 0.1 M sodium acetate. [pKa (CH3COOH) = 4.74 ] Calculate pH of a buffer prepared by adding 10 mL of 0.10 M acetic acid to 20 mL of 0.1 M sodium acetate. [pKa (CH3COOH) = 4.74 ]](https://i.ytimg.com/vi/t9B5VgPOTG4/maxresdefault.jpg)
Calculate pH of a buffer prepared by adding 10 mL of 0.10 M acetic acid to 20 mL of 0.1 M sodium acetate. [pKa (CH3COOH) = 4.74 ]

SOLVED: Question 12 (1 point) What reaction occurs as HCl solution is added to a buffer solution containing equal concentration of acetic acid, CH3COOH; and sodium acetate, CH3COONa? CHzcoo CHzCOoH CHzCOOH Ht

You have 250mL of a 0.56M solution of sodium acetate. How many mL of 0.50M acetic acid should be added to make a buffer of pH 4.40? | Homework.Study.com

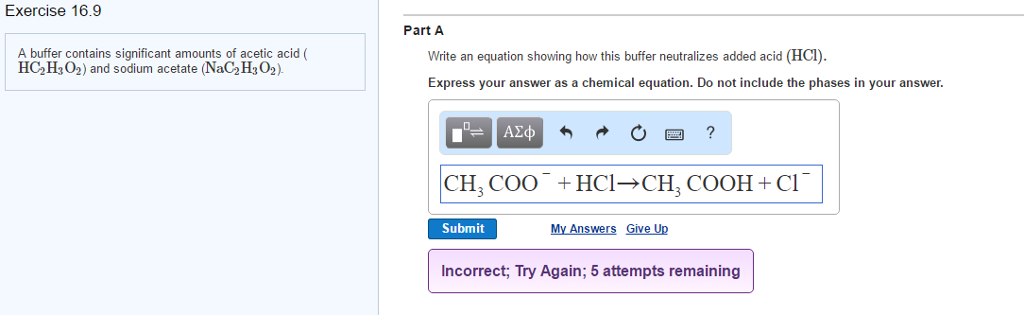
OneClass: A buffer contains significant amounts of acetic acid and sodium acetate. Write an equation ...